PGT-A Testing
Download a PDF of this page:
Introduction
LCRH may advocate PGT-A testing as it is proven to:
Reduce the miscarriage rate and the risk of aneuploid pregnancy and birth.
Reduce the number of unsuccessful embryo transfers therefore reducing the time taken and expense incurred to achieve pregnancy.
Reduce the storage and storage cost of non-viable embryos
Provide answers in the wider fertility investigation
Background
PGT-A (pre-implantation genetic testing for aneuploidy) is a technique used to try and determine whether the embryo tested contains the correct amount of genetic information. Our genetic information is tightly wound and stored within our cells in the form of chromosomes. Usually, we each have 23 pairs of chromosomes: one from our biological mother and one from our biological father for each chromosome. Pairs 1-22 contain the same information in males and females, and pair number 23 determine whether an individual is biologically male (XY) or female (XX). These chromosomes contain genes, which are instructions for our bodies to grow and function.

When one copy of a chromosome is missing it is referred to as a ‘monosomy’ and when there is an extra copy it is called a ‘trisomy’.
The purpose of PGT-A testing is to identify whether embryos have any missing or extra chromosomal information that might result in failed implantation, miscarriage, or the birth of a child with a chromosome abnormality.
An embryo that is created may have more or less than 46 chromosomes which is called aneuploidy. These embryos may fail to develop properly but may also appear to develop quite normally in the laboratory. If an aneuploid embryo is transferred, then there are three potential outcomes:
the embryo fails to develop further and implant
the embryo implants but its development fails in early pregnancy and leads to miscarriage
the embryo implants and develops as if it is a normal pregnancy but is aneuploid
Some aneuploidies can develop to term but may be affected by a serious condition such as Downs Syndrome (trisomy 21), Patau syndrome (trisomy 13), Edwards syndrome (trisomy 18) or Turners syndrome where there is a single X sex chromosome (XO).
What causes aneuploidy?
At birth the ovaries contain all the eggs that will ever be present in a woman’s lifetime. At this stage each egg contains twice the amount of genetic information and divisions need to occur so that a single copy of each chromosome is contained in the egg. As menstrual cycles commence this process of division also commences but errors can occur. It is errors in this division process - called meiosis - that lead to aneuploidies.
As a woman gets older these divisions become less reliable and more aneuploid eggs are produced. This accounts for the lower pregnancy rates and increased miscarriage rates we see with increasing female age.
Approximately 30% of the blastocysts (a fertilized egg after day 5 or 6) created using the eggs of a person aged 35-36 are aneuploid. This rises to 92% when the eggs of a person aged 44 are used to create embryos.
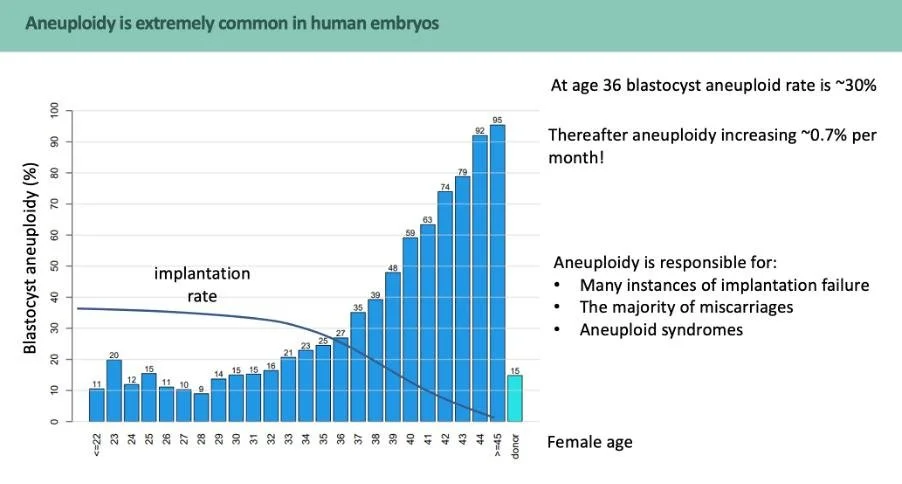
How can we check if an embryo is aneuploid?
Unfortunately, we can’t tell whether an embryo is aneuploid or not by looking at it down a microscope. The only way currently to try and assess the number of chromosomes an embryo contains is to remove some of those cells and analyse the genetic material inside.
What is PGT-A and how is it performed?
Following fertilisation, an embryo divides rapidly over a period of a few days. After 5 or 6 days of development in the laboratory, an embryo contains over 100 cells. At this stage the embryo is known as a blastocyst and it has an outer layer of cells called the trophectoderm which surrounds the outside of the embryo and develops into the placenta. There is also a group of cells inside the embryo called the inner cell mass and it is these cells that develop into the baby. In PGT-A, approximately 5-10 cells are removed from the outer layer of the blastocyst. This procedure, called a biopsy, is performed by a highly skilled senior embryologist. Biopsied embryos are then cryopreserved (frozen) and the biopsied cells are sent to a specialist laboratory for PGT-A analysis.
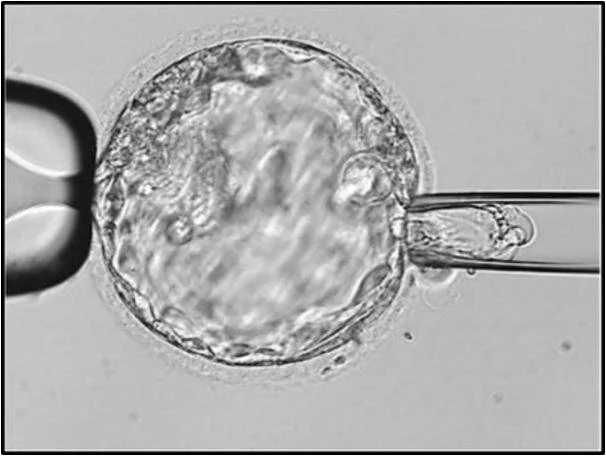
The results of the PGT-A test are available within a few weeks. If the biopsied cells have 46 chromosomes, the embryo they came from is considered euploid/non-aneuploidy detected and the transfer of these embryos can take place in a frozen embryo transfer cycle. Where the biopsied cells had fewer or more that 46 chromosomes, the embryo they came from is considered aneuploid and the embryos are not used in treatment. Occasionally, the results are more complex to interpret and, in these cases access to expert genetic counselling services is available to help patients decide whether to transfer an embryo or not.
Sometimes embryos contain a mixture of euploid and aneuploid cells, which is referred to as mosaicism. Mosaicism means that there are two or more distinct types of cells detected in the embryo. Some cells may have a normal set of chromosomes whilst others are abnormal. If this is occurring in your particular case, at this embryo stage we are unable to clarify the chromosome status of the inner cell mass (the cells which would eventually grow into the foetus). Scientific data about mosaicism suggests that embryos that are reported to be mosaic generally have a similar reproductive potential as euploid/no aneuploidy detected embryos. Published data confirms that embryos classified as ‘abnormal’ have extremely low potential for producing a baby (Tiegs et al., 2021).
Outcomes of PGT-A testing
There are five potential outcomes from PGT-A testing
euploid
aneuploid
segmental aneuploidy
mosaic
no result/non-informative
The best outcome of PGT-A testing is that no aneuploidy is detected. Such embryos are prioritised for transfer. Aneuploid embryos are not transferred and, with consent, are not kept. A mosaic embryo is one where the embryo contains a mixture of normal and abnormal cells. These embryos may be transferred after careful consideration and genetic counselling. Recent studies have demonstrated that many embryos with a mosaic biopsy specimen have similar clinical outcomes to embryos with a euploid result (Capalbo et al., 2021). This study concluded that PGT[A]Seq is >98% accurate in screening for whole chromosome aneuploidy.
Finally, a ‘no result’ is where a sample of cells have been taken for analysis but amplification of the genetic material within those cells has failed to give a result regarding the genetic make-up of those cells. These embryos can be warmed and re-tested.
Benefits of PGT-A
The transfer of chromosomally normal embryos is expected to significantly reduce the frequency of miscarriage and of aneuploid pregnancy, although some risk of these occurrences remains. It is important for all patients to understand that while risks such as an early spontaneous miscarriage or an aneuploid conception may be reduced, they cannot be eliminated. Furthermore, it is important that patients appreciate that PGT-A is not a replacement for routine prenatal testing. It is recommended that prenatal testing should still be undertaken when indicated.
PGT-A testing can help identify the embryo with the best chance of developing into a baby. PGT-A testing can’t change the number of viable embryos developed in a treatment cycle but by transferring only euploid embryos, the time taken to establish a pregnancy could be shorter and the emotional impact of failed cycles and risks of miscarriage could be reduced.
Where patients have undergone multiple rounds of IVF and have no explanation for the treatment failure, PGT-A testing can often provide answers and help with decisions about future treatment options.
If a normal embryo can be identified, then by transferring euploid embryos clinical pregnancy rates are increased and miscarriage rates are decreased.
PGT-A and blastocyst biopsy - things to consider
Some genetic changes cannot be identified using PGT-A, including smaller chromosomal changes and changes within single genes. Additionally, in any pregnancy there is a small chance of a new sporadic genetic condition occurring, which PGT-A cannot detect. If there are any structural rearrangements, PGT-A testing will not detect this as it can only identify the chromosome copy number, not the orientation/arrangement in which the chromosome information is in.
Although PGT-A testing methods are generally very accurate, the test may miss an abnormality or detect one that isn’t there. It is estimated that PGT-A testing will result in a false positive or false negative result in 1-2% of cases. In the case of a false positive, an embryo that may have implanted and developed may be discarded. In the case of a false negative, an aneuploid embryo may be transferred. For this reason, we would always recommend that routine antenatal testing is undertaken even when a euploid embryo has been transferred.
We will not transfer aneuploid embryos.
There is a possible but unquantified risk that removing cells from an embryo may damage it. However, the techniques used for embryo biopsy have been used for many years and there is no suggestion that this is the case. Survival rate post-testing and post-warming is comparable to the one untested embryos exhibit, typically >97%.
Chromosome abnormalities can also arise spontaneously as embryos divide. Where this happens early in development, some cells of the embryo may be chromosomally normal and others abnormal.
As with any treatment there can be financial and emotional costs where treatment including PGT-A is not successful.
Is it possible to choose the sex of my baby?
Although the test does identify the presence of sex chromosomes of the embryo, in the UK it is currently not permitted to select which embryo to transfer based on the sex. For this reason, you won’t know or be able to select the sex of any embryo that is transferred unless you carry a specific sex-linked genetic condition which needs to be avoided. The selection is made by the embryologist solely based on the PGT-A test result and then secondary to this, embryo quality.
Limitations of the PGT-A test
The PGT-A testing method cannot reliably detect abnormalities involving the loss or gain of an entire extra set of chromosomes (the presence of 69 chromosomes or 92 chromosomes).
Sometimes, PGT-A can detect large deletions and duplications of sections of chromosomes (known as segmental aneuploidies), but the clinical significance of these is hard to predict. We can provide access to expert advice in cases where the test results require interpretation.
The test cannot detect small changes in genetic information contained in chromosomes (genes) and the risks of a child inheriting a condition caused by mutation in a single gene (like cystic fibrosis or sickle cell disease, for example) remain the same after PGT-A. There is a separate test looking for more common inherited conditions (see Carrier Screening).
Because testing can take some time, embryos have to be frozen and then thawed and transferred in a frozen embryo transfer cycle. Freezing by vitrification is very effective and 97% of all embryos (biopsied and non-biopsied) survive this process. However, there is a small risk that a frozen embryo will not survive.
Only embryos at the blastocyst stage are suitable for biopsy. If no embryos develop to the blastocyst stage, the test cannot be carried out.
Biopsied cells are couriered to the diagnostic laboratory (Juno Genetics) for PGT-A analysis. Although it’s considered unlikely, it is possible that events or circumstances beyond our control could delay or prevent the delivery of the sample. In this case embryos could be re-biopsied or transferred as in a non PGT-A treatment cycle.
PGT-A and the HFEA
The HFEA have published information regarding optional additional treatments, that they call ‘add-ons’, that clinics offer in addition to the routine treatments they provide.
To make it easier to understand the scientific evidence for each treatment add-on the HFEA developed a rating system for add-ons. There are five ratings that indicate whether a treatment add-on is effective at improving treatment outcomes for someone undergoing fertility treatment, according to evidence from studies. To make it easier to understand their interpretation of the scientific evidence for each treatment add-on the HFEA have devised a range of symbols and colours for each add-on which they have rated.
More information can be found at: https://www.hfea.gov.uk/treatments/treatment-add-ons/
The HFEA has given PGT-A the following ratings on their updated rating system
Rated red for increasing chances of having a baby for most fertility patients
Rated green for reducing the chances of miscarriage for most fertility patients
HFEA Summary of Evidence
For most fertility patients, the use of PGT-A is rated red for improving the chances of having a baby. This is because PGT-A is a selection tool that often reduces the number of embryos available for transfer. In addition, the time to conception resulting in live birth may also be longer.
On balance, findings from high quality evidence shows this add-on is effective at reducing the chances of miscarriage for most fertility patients. PGT-A can be considered where appropriate on an individual basis depending on a patient's personal circumstances and medical history.
This does not remove the chance of having a miscarriage entirely, as there are other reasons a miscarriage may occur other than aneuploidy. The NHS page on miscarriage has further information on this.
For some patients, PGT-A may shorten the time to pregnancy (by avoiding a series of embryo transfers of untested aneuploid embryos).
What does PGT-A cost?
Testing involves a set-up cost and a cost per embryo tested. The set-up cost is higher if the biopsy is being performed on embryos that are already frozen as they must first be thawed, then biopsied, then refrozen while the test results are pending. Please see the LCRH price lists for further information.
Further Support:
Juno Genetics: https://www.junogenetics.co.uk
Fertility Network UK: https://fertilitynetworkuk.org/
The charity UNIQUE : https://rarechromo.org/
Further Reading:
Tiegs AW, Tao X, Zhan Y, Whitehead C, Kim J, Hanson B, Osman E, Kim TJ, Patounakis G, Gutmann J, Castelbaum A, Seli E, Jalas C, Scott RT Jr.
A multicenter, prospective, blinded, nonselection study evaluating the predictive value of an aneuploid diagnosis using a targeted next-generation sequencing-based preimplantation genetic testing for aneuploidy assay and impact of biopsy. Fertil Steril. 2021 Mar;115(3):627-637. doi: 10.1016/j.fertnstert.2020.07.052. Epub 2020 Aug 28. PMID: 32863013
Besser, A., Weidenbaum, E., Buldo-Licciardi, J. et al.
Healthy live births achieved from embryos diagnosed as non-mosaic segmental aneuploid. J Assist Reprod Genet (2024). https://doi.org/10.1007/s10815- 024-03282-8
Kim J, Tao X, Cheng M, Steward A, Guo V, Zhan Y, Scott RT Jr, Jalas C.
The concordance rates of an initial trophectoderm biopsy with the rest of the embryo using PGTseq, a targeted next-generation sequencing platform for preimplantation genetic testing-aneuploidy. Fertil Steril. 2022 Feb;117(2):315-323.

